Ligand screening with NMR
To characterize the binding between ligands (small molecules) and macromolecules (proteins, polymers, nanoparticles, to name a few) is extremely important to many scientific research fields, including medicinal chemistry, drug discovery, material science, and nanotechnology. NMR is a powerful technique to answer the binding or nonbinding question.
This can be done in two ways. The first is receptor (macromolecules) observed methods by following changes in the receptor chemical shifts. The underlining principle is to observe and compare chemical shifts of receptor in the absence and presence of ligand. In particular for proteins, one can isotopically enrich the macromolecule with 13C and/or 15N and collect the HSQC NMR spectrum upon binding of ligands. It's called structure-activity relationship (SAR) by NMR. The second one is to monitor ligands changes with NMR upon binding of macromolecule. There are a few techniques in this category. Some of most popular ones are saturation transfer difference (STD) NMR and Water-Ligand Observed via Gradient SpectroscopY (WaterLOGSY).
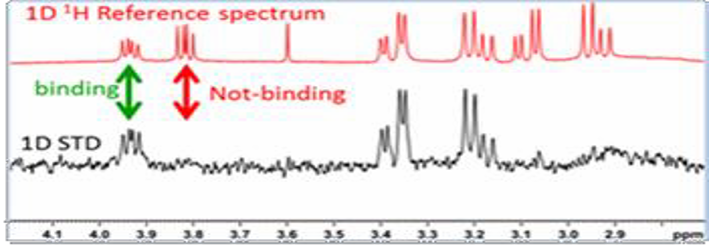
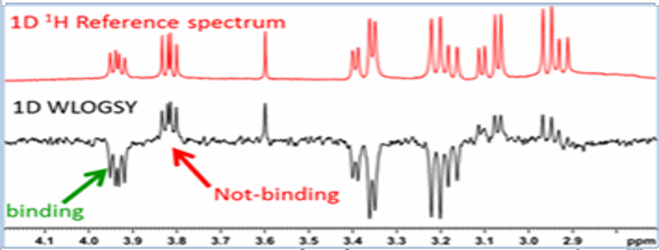
The STD experiment (Mayer, M. and Meyer, B. 1999; Angew. Chem. Int. Ed., 38, 1784-1788.)is performed by selectively saturating a resonance that unique to the receptor only while absent to ligand. If ligand binds, saturation will propagate from the excited protons to other protons of receptor through spin diffusion. The same saturation is further transferred to binding ligands by cross relaxation at the binding interface. A control experiment is done by saturating a region of the spectrum that does not contain signal. The resulting difference spectrum yields only those resonances that have experienced saturation, i.e. those from the compound that binds to the receptor. The receptor is typically present at very small concentrations so its resonances will not be visible. The way to differentiate binders from non-binders is showed in the picture below: binders show up in difference spectrum while the non-binders don’t:WaterLOGSY (Dalvit C, etc. J Biomol NMR. 2001;21:349–359.) is a variant of STD where saturation transfer involves bound water instead of protein i.e. saturate water resonance. Magnetization between water and protein is transferred through intermolecular NOE and spin diffusion. The ligands interact with water via water–ligand–protein or protein–ligand complexes, whose rotational correlation times yield negative cross-relaxation rates and exhibit a negative NOE with water. By contrast, small molecules that only interact with bulk water (non-binders) will experience much faster tumbling, which translates into a positive NOE. Therefore, opposite signs for signals from free versus protein-bound ligands are observed in a WaterLOGSY spectrum, which enables one to easily discriminate binders and non-binders. As you can see from below figure, binders have opposite phase to non-binders:
You can run all these experiments on HFCN600 after trained by staff. The NMR staff also provide service on SAR by NMR, STD NMR, and WaterLOGSY. Please click to start a project.