Powder Diffraction
Overview
Powder diffraction (PXRD) is a rapid technique that can be used for phase identification and purity analysis of bulk crystalline materials. Combined with elemental analysis , PXRD methods can be used for the identification of unknown samples and/or impurities. Additionally, when single crystal growth is challenging, powder diffraction methods can be utilized for indexing to solving the structure from diffraction data collected on powders. Furthermore, PXRD can be combined with a variety of in-situ/operando methods for measurements under non-ambient conditions. Please check out some selected publications resulted by IMSERC users along with the list of other crystallographic services available in IMSERC
Applications
- Determination and refinement of unit cell
- Powder evaluation of sample purity (sensitivity of ~2% by weight)
- Quantitative determination of individual crystalline phases and impurities in mixtures of powder
- Monitor reactions in real time as a function of time, temperature, pressure, and gas flow/pressure
- Probe catalytic changes to substrates
- Investigate decomposition mechanism
- Crystalline particle size determination
- In-situ monitoring of crystallization processes with increasing temperature
- Construction of phase diagrams
- Thermal expansion coefficient measurement
- Rocking curve measurements for evaluation of defect density and quality of crystals
- Texture measurements and orientation of grains in a polycrystalline sample
- Strain analysis
Please visit our expanded list of application per research area for more details about this technique.
If you are interested in utilizing any of these applications for your research Start a project
Selected Publications
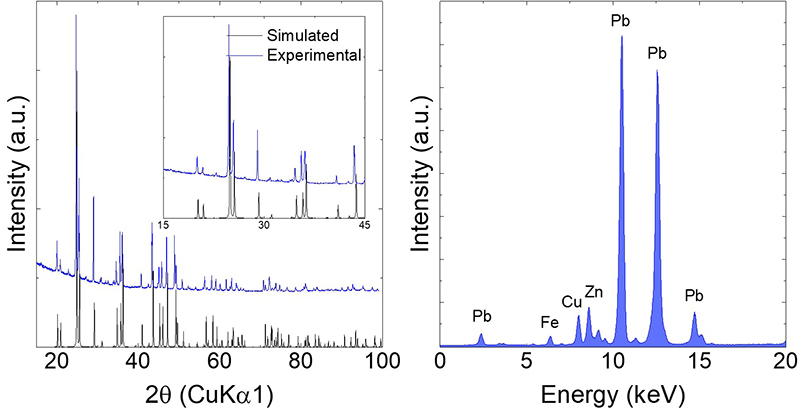
Structure solution from variable temperature powder diffraction data
Ag2Se to KAg3Se2: Suppressing Order-Disorder Transitions via Reduced Dimensionality
Rettie, A. J. E.; Malliakas, C. D.; Botana, A. S.; Hodges, J. M.; Han, F.; Huang, R.; Chung, D. Y.; Kanatzidis, M. G. [10.1021/jacs.8b04888]
Strain analysis
Lattice Softening Significantly Reduces Thermal Conductivity and Leads to High Thermoelectric Efficiency
Hanus, R.; Agne, M. T.; Rettie, A. J. E.; Chen, Z.; Tan, G.; Chung, D. Y.; Kanatzidis, M. G.; Pei, Y.; Voorhees, P. W.; Snyder, G. J. [10.1002/adma.201900108]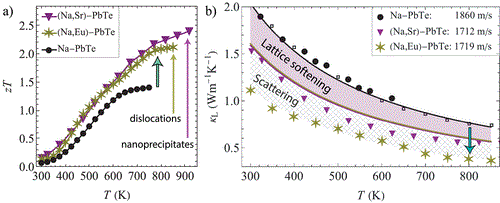
Texture analysis
Anisotropic Redox Conductivity within a Metal-Organic Framework Material
Goswami, S.; Hod, I.; Duan, J. D.; Kung, C. W.; Rimoldi, M.; Malliakas, C. D.; Palmer, R. H.; Farha, O. K.; Hupp, J. T. [10.1021/jacs.9b07658]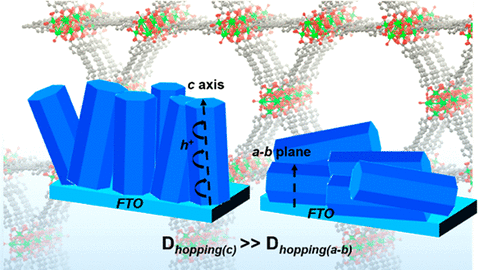
Structure of Magnetic 2D Metal-Organic Frameworks
Synthetic investigation of competing magnetic interactions in 2D metal-chloranilate radical frameworks
Collins, K. A.; Saballos, R. J.; Fataftah, M. S.; Puggioni, D.; Rondinelli, J. M.; Freedman, D. E. [10.1039/d0sc01994a]