
 Through-bond assignment of sugar 1H
and 13C resonances
Through-bond assignment of sugar 1H
and 13C resonances
| Proton |
Range 1H
chemical shift |
Range 13C
chemical shift |
|
1'
|
5.4-6.4
|
82.0-87.0
|
|
2'
|
2.0-3.0
|
38.0-41.0
|
|
2''
|
-
|
-
|
|
3'
|
4.5-5.2
|
70.0-79.0
|
|
4'
|
4.0-4.5
|
83.0-88.0
|
|
5'
|
3.6-4.4
|
61.0-68.0
|
|
5''
|
3.6-4.4
|
61.0-68.0
|
- Assignment of non-exchangeable sugar protons
via NOE from 2D NOESY experiment and via
J-coupling from 2D COSY and
2D TOCSY experiments.
Overlapping can be resolved using hybrid experiments like 3D
TOCSY-NOESY and 3D NOESY-NOESY
-
Directly-attached 1H-13C pairs are assigned from
standard 2D 1H-13C HMQC/
HSQC
experiments.
-
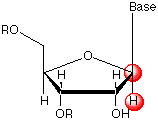
Stereospecific H5' and H5" proton assignments can be achieved from long-range proton-carbon
coupling constants analysis extracted from multi-dimensional
HMQC-TOCSY
(
93JACS11002
and
94JACS5823
)
or HMQC-NOESY
(
94JACS5823
)
experiments.Analysis of cross-peak signal intensities
of C3'-H5 in 2D HMBC spectra can also be helpful
-
Stereospecific H2' and H2" proton assignments in deoxyribose can be achieved from
intraresidue di(1':2',2") NOE cross-peak analysis
or from the measurement of 3J1'2' and 3J1'2"
coupling constants.